OLIMEL N7E Emulsion for Infusion
4.1 Therapeutic indications
OLIMEL N7E is indicated for parenteral nutrition for adults and children greater than 2 years of age when oral or enteral nutrition is impossible, insufficient or contraindicated.
4.3 Contraindications
The administration of OLIMEL N7E is contraindicated in the following situations:
- In premature neonates, infants, and children less than 2 years of age,
- Hypersensitivity to egg, soya-bean, peanut proteins, or corn/corn products (see section 4.4 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information) or to any of the active substances or excipients, listed in section 6.1 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information,
- Congenital abnormalities of amino acid metabolism,
- Severe hyperlipidaemia or severe disorders of lipid metabolism characterised by hypertriglyceridaemia,
- Severe hyperglycaemia,
- Pathologically-elevated plasma concentrations of sodium, potassium, magnesium, calcium and/or phosphorus.
4.2 Posology and method of administration
Posology
OLIMEL N7E is not recommended for use in children less than 2 years of age due to inadequate composition and volume (see sections 4.4; 5.1 and 5.2 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information).
The maximum daily dose mentioned below should not be exceeded. Due to the static composition of the multi-chamber bag, the ability to simultaneously meet all nutrient needs of the patient may not be possible. Clinical situations may exist where patients require amounts of nutrients varying from the composition of the static bag. In this situation any volume (dose) adjustments must take into consideration the resultant effect this will have on the dosing of all other nutrient components of OLIMEL N7E.
In adults
The dosage depends on the patient’s energy expenditure, clinical status, body weight, and the ability to metabolise the constituents of OLIMEL N7E, as well as additional energy or proteins provided orally/enterally; therefore, the bag size should be chosen accordingly.
The average daily requirements are:
- 0.16 to 0.35 g nitrogen per kg body weight (1 to 2 g of amino acids/kg), depending on the patient's nutritional status and degree of catabolic stress,
- 20 to 40 kcal/kg,
- 20 to 40 ml fluid /kg, or 1 to 1.5 ml per expended kcal.
For OLIMEL N7E, the maximal daily dose is defined by total caloric intake, 40 kcal/kg provided in a volume of 35 ml/kg, corresponding to 1.5 g/kg amino acids, 4.9 g/kg glucose and 1.4 g/kg lipids, 1.2 mmol/kg sodium, and 1.1 mmol/kg potassium. For a 70 kg patient, this would be equivalent to 2450 ml OLIMEL N7E per day, resulting in an intake of 108 g amino acids, 343 g glucose and 98 g lipids (i.e. 2352 non-protein kcal and 2 793 total kcal).
Normally, the flow rate must be increased gradually during the first hour and then be adjusted to take into account the dose being administered, the daily volume intake and the duration of the infusion.
For OLIMEL N7E, the maximal infusion rate is 1.7 ml/kg/hour, corresponding to 0.08 g/kg/hour amino acids, 0.24 g/kg/hour glucose, and 0.07 g/kg/hour lipids.
In children greater than 2 years of age and adolescents
There have been no studies performed in the paediatric population.
The dosage depends on the patient’s energy expenditure, clinical status, body weight, and the ability to metabolise constituents of OLIMEL N7E, as well as additional energy or proteins given orally/enterally; therefore, the bag size should be chosen accordingly.
In addition, daily fluid, nitrogen and energy requirements continuously decrease with age. Two groups, ages 2 to 11 years and 12 to 18 years, are considered:
For OLIMEL N7E, in both age groups, the magnesium concentration is the limiting factor for daily dose. In both age groups, the glucose concentration is the limiting factor for hourly rate. The resulting intakes are displayed below:
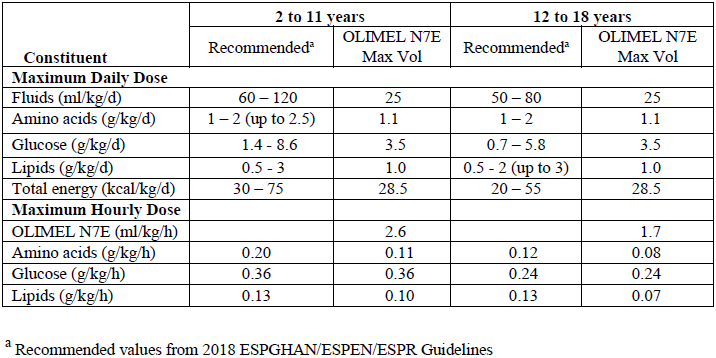
Normally, the flow rate must be increased gradually during the first hour and then be adjusted to take into account the dose being administered, the daily volume intake, and the duration of the infusion.
In general, it is recommended to start the infusion for small children with low daily dose and gradually increase it up to the maximal dosage (see above).
Method and duration of administration
For single use only.
It is recommended that, after opening the bag, the contents are used immediately and not stored for subsequent infusion.
After reconstitution, the mixture is homogenous with a milky appearance.
For instructions for preparation and handling of the emulsion for infusion see section 6.6 – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
Due to its high osmolarity, OLIMEL N7E can only be administered through a central vein.
The recommended duration of infusion for a parenteral nutrition bag is between 12 and 24 hours.
Treatment with parenteral nutrition may be continued for as long as required by the patient’s clinical conditions.