PLUVICTO SOLUTION FOR INJECTION/INFUSION 1000 MBq/mL
3 Indications
Pluvicto® is indicated for the treatment of adult patients with prostate-specific membrane antigen (PSMA)-positive metastatic castration-resistant prostate cancer (mCRPC) who have been treated with androgen receptor (AR) pathway inhibitor and taxane-based chemotherapy.
5 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in Section 2 Description and composition – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information.
4 Dosage regimen and administration
Important safety instructions
Pluvicto is a radiopharmaceutical and should be handled with appropriate safety measures to minimize radiation exposure (see section 6 Warnings and precautions – please refer to the Product Insert/Patient Information Leaflet published on HSA for the full drug information). Waterproof gloves and effective radiation shielding should be used when handling Pluvicto.
Radiopharmaceuticals, including Pluvicto, should be used by or under the control of healthcare providers who are qualified by specific training and experience in the safe use and handling of radiopharmaceuticals.
Patient identification
Patients should be identified for treatment by PSMA imaging.
Dosage regimen
The recommended Pluvicto dose is 7.4 GBq (200 mCi) intravenously every 6 weeks (± 1 week) for a total of 6 doses.
Treatment monitoring
Laboratory tests should be performed before and during treatment with Pluvicto.
- Hematology (hemoglobin, white blood cell count, absolute neutrophil count, platelet count)
- Kidney function (serum creatinine, calculated creatinine clearance [CLcr])
- Liver function (alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, blood serum albumin, total blood bilirubin)
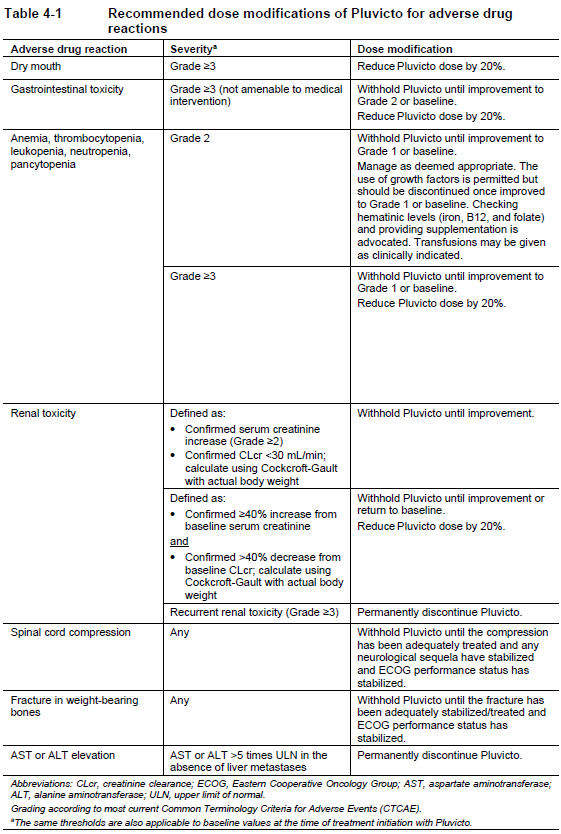
Dose modifications for adverse drug reactions
Recommended dose modifications of Pluvicto for adverse drug reactions are provided in Table 4-1. Management of severe or intolerable adverse drug reactions may require temporary dose interruption (extending the dosing interval by 4 weeks from 6 weeks up to 10 weeks), dose reduction or permanent discontinuation of treatment with Pluvicto. If a treatment delay due to an adverse drug reaction persists for >4 weeks, treatment with Pluvicto must be discontinued. The dose of Pluvicto may be reduced by 20% once; the dose should not be re-escalated. If a patient has further adverse drug reactions that would require an additional dose reduction, treatment with Pluvicto must be discontinued.
Special populations
Renal impairment
Exposure of Pluvicto is expected to increase with the degree of renal impairment. Patients with mild or moderate renal impairment may be at greater risk of toxicity. No dose adjustment is recommended for patients with mild renal impairment (baseline CLcr 60 to 89 mL/min by Cockcroft-Gault); however, insufficient data are available for drawing a conclusion for patients with moderate renal impairment (CLcr 30 to 59 mL/min). Renal function and adverse reactions should be monitored frequently in patients with mild to moderate renal impairment. The pharmacokinetic profile and safety of Pluvicto have not been studied in patients with severe (CLcr 15 to 29 mL/min) renal impairment or end-stage renal disease.
Hepatic impairment
No dose adjustment is recommended for patients with hepatic impairment.
Pediatric patients (below 18 years of age)
The safety and effectiveness of Pluvicto in pediatric patients have not been established.
Geriatric patients (65 years of age or older)
No dose adjustment is recommended in patients 65 years or older.
Method of administration
Preparation instructions
- Aseptic technique and radiation shielding should be used when handling or administering Pluvicto, using tongs as needed to minimize radiation exposure.
- The vial should be visually inspected under a shielded screen for particulate matter and discoloration prior to administration. The vial should be discarded if particulates or discoloration are present.
- Pluvicto is a ready-to-use solution for single use only. The Pluvicto solution should not be injected directly into any other intravenous solution.
- The amount of radioactivity delivered to the patient should be confirmed with an appropriately calibrated dose calibrator prior to and after Pluvicto administration.
- Any unused medicinal product or waste material should be disposed of in accordance with national regulations.
Administration instructions
The recommended dose of Pluvicto may be administered intravenously as an injection using a disposable syringe fitted with a syringe shield (with or without a syringe pump), as an infusion using the gravity method (with or without an infusion pump), or as an infusion using the vial (with a peristaltic infusion pump).
A reduced dose of Pluvicto should be administered using the syringe method (with or without a syringe pump) or the vial method (with a peristaltic infusion pump). Using the gravity method to administer a reduced dose of Pluvicto is not recommended since it may result in delivery of the incorrect volume of Pluvicto if the dose is not adjusted prior to administration.
Prior to administration, flush the intravenous catheter used exclusively for Pluvicto administration with ≥10 mL of 0.9% sterile sodium chloride solution to ensure patency and to minimize the risk of extravasation. Cases of extravasation should be managed as per institutional guidelines.
Intravenous methods of administration
Instructions for the syringe method (with or without a syringe pump)
- After disinfecting the vial stopper, withdraw an appropriate volume of Pluvicto solution to deliver the desired radioactivity by using a disposable syringe fitted with a syringe shield and a disposable sterile needle.
- Administer Pluvicto to the patient by slow intravenous push within approximately 1 to 10 minutes (either with a syringe pump or manually without a syringe pump) via an intravenous catheter that is pre-filled with 0.9% sterile sodium chloride solution and that is used exclusively for Pluvicto administration to the patient.
- Once the desired Pluvicto radioactivity has been administered, perform an intravenous flush of ≥10 mL of 0.9% sterile sodium chloride solution through the intravenous catheter to the patient.
Instructions for the gravity method (with or without an infusion pump)
- Insert a 2.5 cm, 20 gauge needle (short needle) into the Pluvicto vial and connect via a catheter to 500 mL 0.9% sterile sodium chloride solution (used to transport the Pluvicto solution during the infusion). Ensure that the short needle does not touch the Pluvicto solution in the vial and do not connect the short needle directly to the patient. Do not allow the sodium chloride solution to flow into the Pluvicto vial prior to the initiation of the Pluvicto infusion and do not inject the Pluvicto solution directly into the sodium chloride solution.
- Insert a second needle that is 9 cm, 18 gauge (long needle) into the Pluvicto vial, ensuring that the long needle touches and is secured to the bottom of the Pluvicto vial during the entire infusion. Connect the long needle to the patient by an intravenous catheter that is pre-filled with 0.9% sterile sodium chloride solution and that is used exclusively for the Pluvicto infusion into the patient.
- Use a clamp or an infusion pump to regulate the flow of the sodium chloride solution via the short needle into the Pluvicto vial (the sodium chloride solution entering the vial through the short needle will carry the Pluvicto solution from the vial to the patient via the intravenous catheter connected to the long needle within approximately 30 minutes).
- During the infusion, ensure that the level of solution in the Pluvicto vial remains constant.
- Disconnect the vial from the long needle line and clamp the saline line once the level of radioactivity is stable for at least five minutes.
- Follow the infusion with an intravenous flush of ≥10 mL of 0.9% sterile sodium chloride solution through the intravenous catheter to the patient.
Instructions for the vial method (with a peristaltic infusion pump)
- Insert a 2.5 cm, 20 gauge needle (short venting needle) into the Pluvicto vial. Ensure that the short needle does not touch the Pluvicto solution in the vial and do not connect the short needle directly to the patient or to the peristaltic infusion pump.
- Insert a second needle that is 9 cm, 18 gauge (long needle) into the Pluvicto vial, ensuring that the long needle touches and is secured to the bottom of the Pluvicto vial during the entire infusion. Connect the long needle and a 0.9% sterile sodium chloride solution to a 3-way stopcock valve via appropriate tubing.
- Connect the output of the 3-way stopcock valve to tubing installed on the input side of the peristaltic infusion pump following the pump manufacturer’s instructions.
- Pre-fill the line by opening the 3-way stopcock valve and pumping the Pluvicto solution through the tubing until it reaches the exit of the valve.
- Pre-fill the intravenous catheter which will be connected to the patient by opening the 3-way stopcock valve to the 0.9% sterile sodium chloride solution and pumping the 0.9% sterile sodium chloride solution until it exits the end of the catheter tubing.
- Connect the pre-filled intravenous catheter to the patient and set the 3-way stopcock valve such that the Pluvicto solution is in line with the peristaltic infusion pump.
- Infuse an appropriate volume of Pluvicto solution at approximately 25 mL/h to deliver the desired radioactivity.
- When the desired Pluvicto radioactivity has been delivered, stop the peristaltic infusion pump and then change the position of the 3-way stopcock valve so that the peristaltic infusion pump is in line with the 0.9% sterile sodium chloride solution. Restart the peristaltic infusion pump and infuse an intravenous flush of ≥10 mL of 0.9% sterile sodium chloride solution through the intravenous catheter to the patient.